The α-substitution reaction of a carbonyl compound through either an enol or enolate ion
intermediate is one of the four fundamental reaction types in carbonyl-group chemistry.
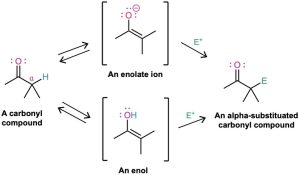
Carbonyl compounds are in an equilibrium with their enols, a process called keto–enol tautomerism. Although enol tautomers are normally present to only a small extent at equilibrium and can’t usually be isolated in pure form, they nevertheless contain a highly nucleophilic double bond and react with electrophiles in an α-substitution reaction. An example is the α halogenation of ketones on treatment with Cl2, Br2, or I2 in acid solution. Alpha bromination of carboxylic acids can be similarly accomplished by the Hell–Volhard– Zelinskii (HVZ) reaction, in which an acid is treated with Br2 and PBr3. The α-halogenated products can then undergo base-induced E2 elimination to yield α,β-unsaturated carbonyl compounds.
Alpha hydrogen atoms of carbonyl compounds are weakly acidic and can be removed by strong bases, such as lithium diisopropylamide (LDA), to yield nucleophilic enolate ions. The most useful reaction of enolate ions is their SN2 alkylation with alkyl halides. The Malonic ester synthesis converts an alkyl halide into a carboxylic acid with the addition of two carbon atoms (RX → RCH2CO2H). Similarly, the acetoacetic ester synthesis converts an alkyl halide into a methyl ketone with the addition of three carbon atoms (RX → RCH2COCH3). In addition, many carbonyl compounds, including ketones, esters, and nitriles, can be directly alkylated by treatment with LDA and an alkyl halide.

