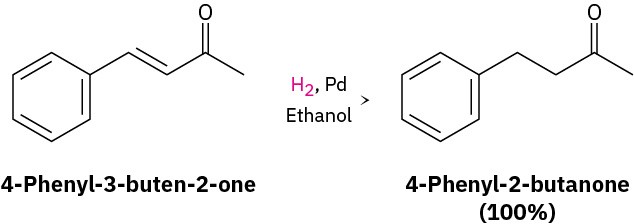
Catalytic Hydrogenation of Aromatic Rings
Just as aromatic rings are generally inert to oxidation, they’re also inert to catalytic hydrogenation under conditions that reduce typical alkene double bonds. As a result, it’s possible to reduce an alkene double bond selectively in the presence of an aromatic ring. For example, 4-phenyl-3-buten-2-one is reduced to 4-phenyl-2-butanone using a palladium catalyst at room temperature and atmospheric pressure. Neither the benzene ring nor the ketone carbonyl group is affected.
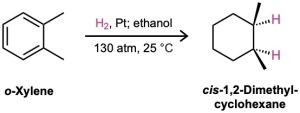
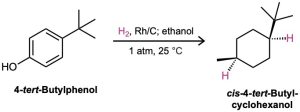
To hydrogenate an aromatic ring, it’s necessary either to use a platinum catalyst with hydrogen gas at a pressure of several hundred atmospheres or to use a more effective catalyst such as rhodium on carbon. Under these conditions, aromatic rings are converted into cyclohexanes. For example, o-xylene yields 1,2-dimethylcyclohexane, and 4-tert– butylphenol gives 4-tert-butylcyclohexanol.
Reduction of Aryl Alkyl Ketones
In the same way that an aromatic ring activates a neighboring (benzylic) C–H toward oxidation, it also activates a benzylic carbonyl group toward reduction. Thus, an aryl alkyl ketone prepared by Friedel–Crafts acylation of an aromatic ring can be converted into an alkylbenzene by catalytic hydrogenation over a palladium catalyst. Propiophenone, for instance, is reduced to propylbenzene by catalytic hydrogenation. Because the net effect of Friedel–Crafts acylation followed by reduction is the preparation of a primary alkylbenzene, this two-step sequence of reactions makes it possible to circumvent the carbocation rearrangement problems associated with direct Friedel–Crafts alkylation using a primary alkyl halide (Section 16.3).
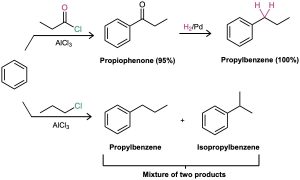
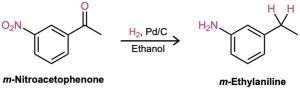
The conversion of a carbonyl group into a methylene group (C=O → CH2) by catalytic hydrogenation is limited to aryl alkyl ketones; dialkyl ketones are not reduced under these conditions. Furthermore, the catalytic reduction of aryl alkyl ketones is not compatible with the presence of a nitro substituent on the aromatic ring because a nitro group is reduced to an amino group under reaction conditions. We’ll see a more general method for reducing ketone carbonyl groups to yield alkanes in Section 19.9.
Problem 16-21
How would you prepare diphenylmethane, (Ph)2CH2, from benzene and an acid chloride?

