The final point we’ll consider about cycloalkane stereochemistry is to see what happens when two or more cycloalkane rings are fused together along a common bond to construct a polycyclic molecule—for example, decalin.

Decalin consists of two cyclohexane rings joined to share two carbon atoms (the bridgehead carbons, C1 and C6) and a common bond. Decalin can exist in either of two isomeric forms, depending on whether the rings are trans fused or cis fused. In cis-decalin, the hydrogen atoms at the bridgehead carbons are on the same side of the rings; in trans– decalin, the bridgehead hydrogens are on opposite sides. Figure 4.18 shows how both compounds can be represented using chair cyclohexane conformations. Note that the two decalin isomers are not interconvertible by ring-flips or other rotations. They are cis–trans stereoisomers and have the same relationship to each other that cis– and trans-1,2-dimethylcyclohexane have.
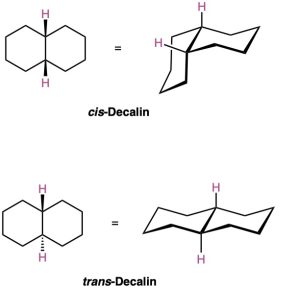
Figure 4.18 Representations of cis– and trans-decalin. Hydrogen atoms at the bridgehead carbons are on the same face of the rings in the cis isomer but on opposite faces in the trans isomer.
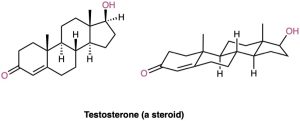
Polycyclic compounds are common in nature, and many valuable substances have fused-ring structures. For example, steroids, such as testosterone, the primary sex hormone in males, have three six-membered rings and one five-membered ring fused together. Although steroids look complicated compared with cyclohexane or decalin, the same principles that apply to the conformational analysis of simple cyclohexane rings apply equally well (and often better) to steroids.

Another common ring system is the norbornane (bicyclo[2.2.1]heptane) structure. Like decalin, norbornane is a bicycloalkane, so called because two rings would have to be broken open to generate an acyclic structure. Its systematic name, bicyclo[2.2.1]heptane, reflects the fact that the molecule has seven carbons, is bicyclic, and has three “bridges” of 2, 2, and 1 carbon atoms connecting the two bridgehead carbons.
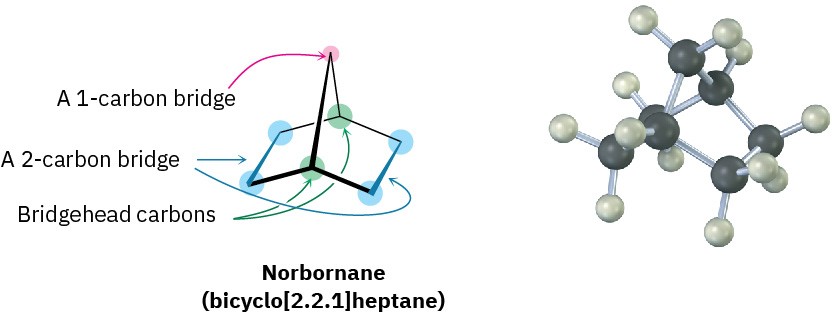
Norbornane has a conformationally locked boat cyclohexane ring in which carbons 1 and 4 are joined by an additional CH2 group. In drawing this structure, a break in the rear bond indicates that the vertical bond crosses in front of it. Making a molecular model is particularly helpful when trying to see the three-dimensionality of norbornane.
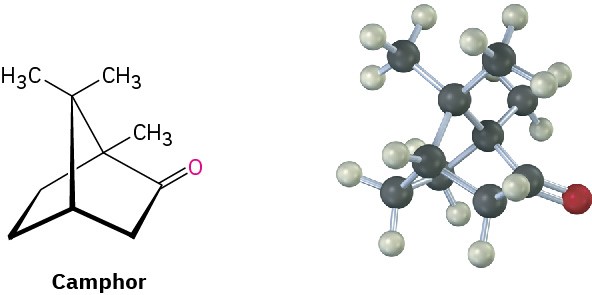
Substituted norbornanes, such as camphor, are found widely in nature, and many have been important historically in developing organic structural theories.
Problem 4-20
Which isomer is more stable, cis-decalin or trans-decalin (Figure 4.18)? Explain.
Problem 4-21
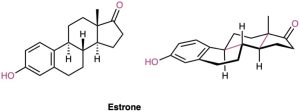
Look at the following structure of estrone, the primary sex hormone in females, and tell whether each of the two indicated (red) ring-fusions is cis or trans.