Most of the biochemical reactions that take place in the body, as well as many organic reactions in the laboratory, yield products with chirality centers. For example, acid- catalyzed addition of H2O to 1-butene in the laboratory yields 2-butanol, a chiral alcohol. What is the stereochemistry of this chiral product? If a single enantiomer is formed, is it R or S? If a mixture of enantiomers is formed, how much of each? In fact, the 2-butanol produced is a racemic mixture of R and S enantiomers. Let’s see why.
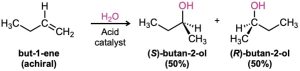
To understand why a racemic product results from the reaction of H2O with 1-butene, think about the reaction mechanism. 1-Butene is first protonated to yield an intermediate secondary carbocation. Because the trivalent carbon is sp2-hybridized and planar, the cation has a plane of symmetry and is achiral. As a result, it can react with H2O equally well from either the top or the bottom. Reaction from the top leads to (S)-2-butanol through transition state 1 (TS 1) in Figure 8.13, and reaction from the bottom leads to (R)-2-butanol through TS 2. But the two transition states are mirror images, so they have identical energies, form at identical rates, and are equally likely to occur.
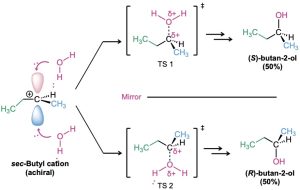
Figure 8.13 Reaction of H2O with the carbocation resulting from protonation of 1- butene. Reaction from the top leads to S product and is the mirror image of reaction from the bottom, which leads to R product. Because they are energetically identical, they are equally likely and lead to a racemic mixture of products. The dotted C···O bond in the transition state indicates partial bond formation.
As a general rule, the formation of a new chirality center by achiral reactants always leads to a racemic mixture of enantiomeric products. Put another way, optical activity can’t appear from nowhere; an optically active product can only result by starting with an optically active reactant or chiral environment (Section 5.12).
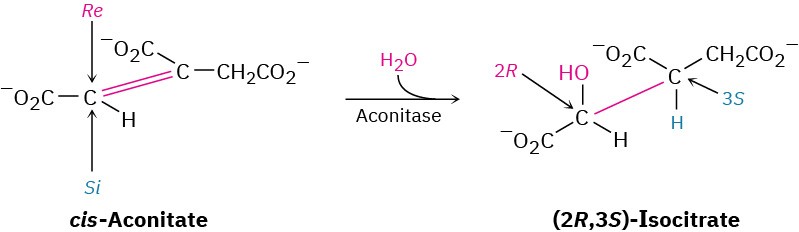
In contrast to laboratory reactions, enzyme-catalyzed biological reactions often give a single enantiomer of a chiral product, even when the substrate is achiral. One step in the citric acid cycle of food metabolism, for instance, is the aconitase-catalyzed addition of water to (Z)-aconitate (usually called cis-aconitate) to give isocitrate.
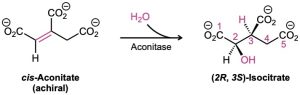
Even though cis-aconitate is achiral, only the (2R,3S) enantiomer of the product is formed. As discussed in Section 5.11 and Section 5.12, cis-aconitate is a prochiral molecule, which is held in a chiral environment by the aconitase enzyme during the reaction. In this environment, the two faces of the double bond are chemically distinct, and addition occurs on only the Re face at C2.