8 Why This Chapter?
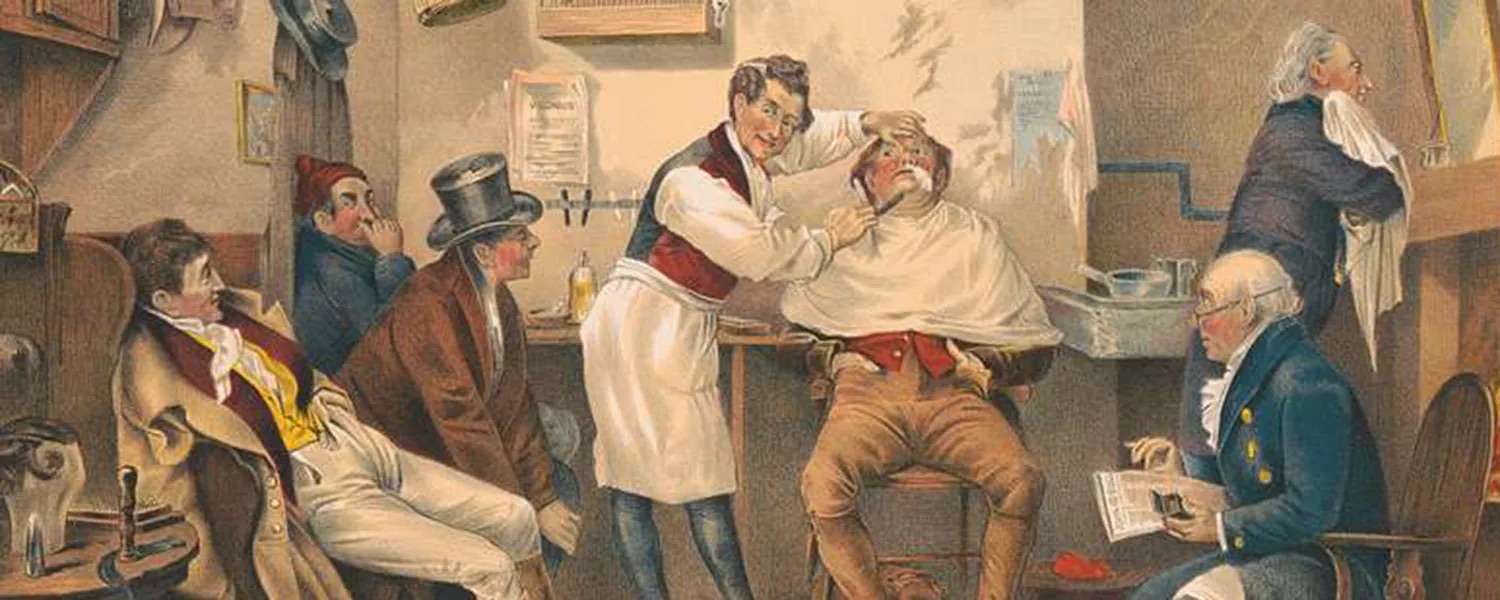
Figure 8.1 In the 19th and early-20th centuries, benzene was used as an aftershave lotion because of its pleasant smell and as a solvent to decaffeinate coffee beans. Neither is a good idea. (credit: modification of work “Ye Old Way” by Geo. H. Walker & Co./Library of Congress)
The reactivity of substituted aromatic compounds, more than that of any other class of substances, is intimately tied to their exact structure. As a result, aromatic compounds provide an extraordinarily sensitive probe for studying the relationship between structure and reactivity. We’ll examine this relationship here and in the following chapter, and we’ll find that the lessons learned are applicable to all other organic compounds, including important substances such as the nucleic acids that control our genetic makeup.
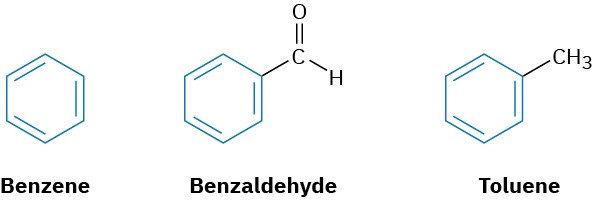
In the early days of organic chemistry, the word aromatic was used to describe such fragrant substances as benzaldehyde (from cherries, peaches, and almonds), toluene (from Tolu balsam), and benzene (from coal distillate). It was soon realized, however, that substances grouped as aromatic differed from most other organic compounds in their chemical behavior.
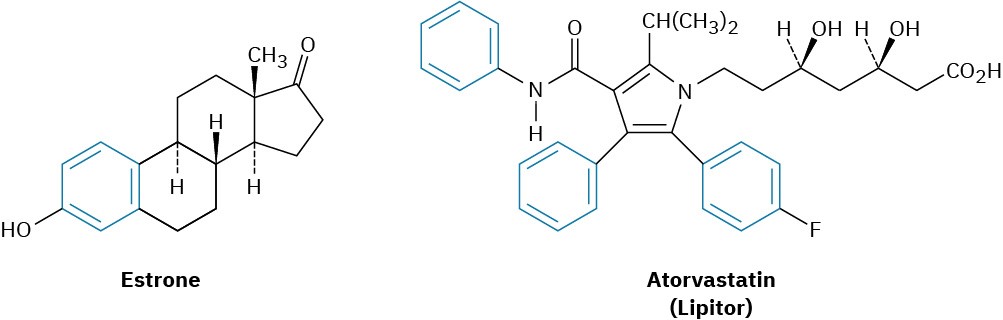
 Today, the association of aromaticity with fragrance has long been lost, and we now use the word aromatic to refer to the class of compounds that contain six-membered rings with three alternating double bonds. Many naturally occurring compounds are aromatic in part, including steroids such as estrone and well-known pharmaceuticals such as the cholesterol-lowering drug atorvastatin, marketed as Lipitor. Benzene itself causes a depressed white blood cell count (leukopenia) on prolonged exposure and should not be used as a laboratory solvent.
Today, the association of aromaticity with fragrance has long been lost, and we now use the word aromatic to refer to the class of compounds that contain six-membered rings with three alternating double bonds. Many naturally occurring compounds are aromatic in part, including steroids such as estrone and well-known pharmaceuticals such as the cholesterol-lowering drug atorvastatin, marketed as Lipitor. Benzene itself causes a depressed white blood cell count (leukopenia) on prolonged exposure and should not be used as a laboratory solvent.
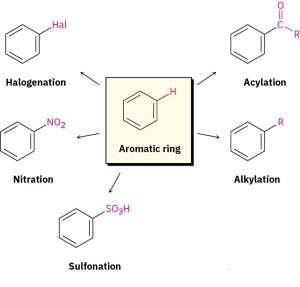
The most common reaction of aromatic compounds is electrophilic aromatic substitution, in which an electrophile (E+) reacts with an aromatic ring and substitutes for one of the hydrogens. The reaction is characteristic of all aromatic rings, not just benzene and substituted benzenes. In fact, the ability of a compound to undergo electrophilic substitution is a good test of aromaticity.
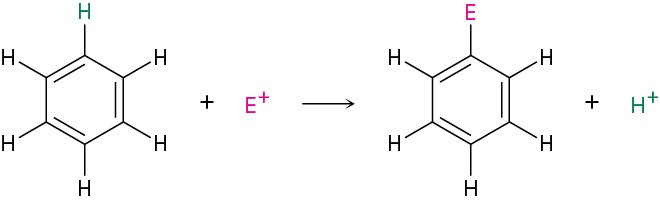 Many different substituents can be introduced onto an aromatic ring through electrophilic substitution. To list some possibilities, an aromatic ring can be substituted by a halogen (–Cl, –Br, –I), a nitro group (–NO2), a sulfonic acid group (–SO3H), an alkyl group (–R), or an acyl group (–COR). Starting from only a few simple materials, it’s possible to prepare many thousands of substituted aromatic compounds.
Many different substituents can be introduced onto an aromatic ring through electrophilic substitution. To list some possibilities, an aromatic ring can be substituted by a halogen (–Cl, –Br, –I), a nitro group (–NO2), a sulfonic acid group (–SO3H), an alkyl group (–R), or an acyl group (–COR). Starting from only a few simple materials, it’s possible to prepare many thousands of substituted aromatic compounds.