9.4 Preparation of Alcohols: A Review
Alcohols occupy a central position in organic chemistry. They can be prepared from many other kinds of compounds (alkenes, alkyl halides, ketones, esters, and aldehydes, among others), and they can be transformed into an equally wide assortment of compounds (Figure 9.5).
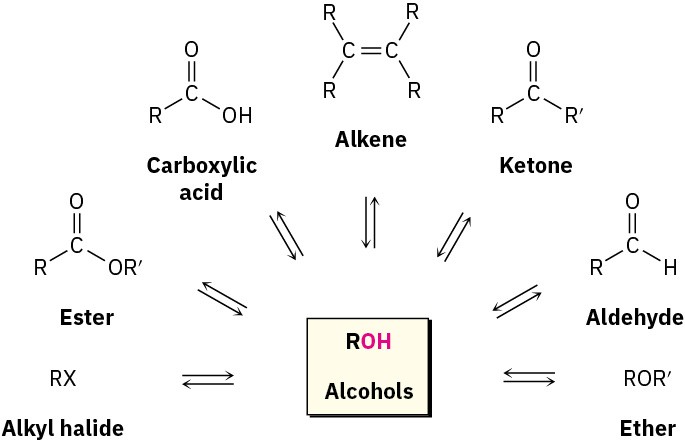 Figure 9.5 The central position of alcohols in organic chemistry. Alcohols can be prepared from, and converted into, many other kinds of compounds.
Figure 9.5 The central position of alcohols in organic chemistry. Alcohols can be prepared from, and converted into, many other kinds of compounds.
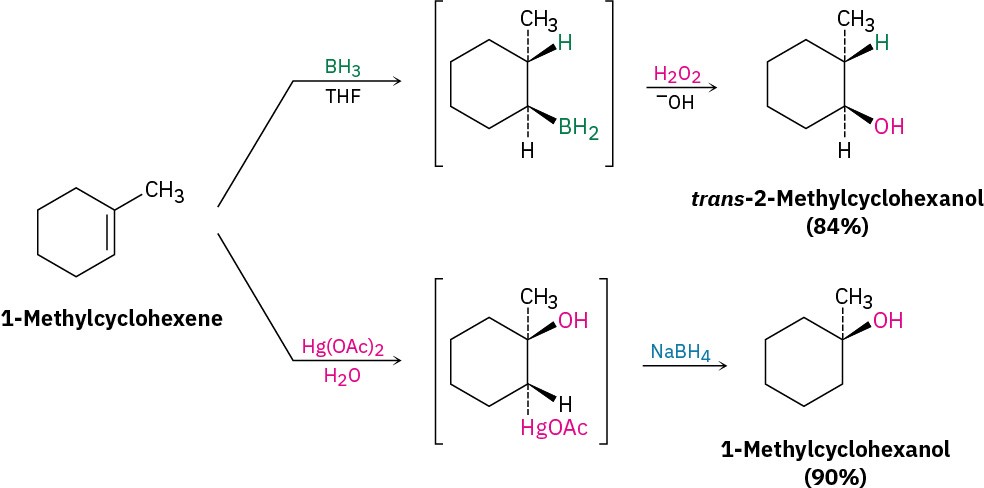
Alcohols can be prepared by hydration of alkenes. Because the direct hydration of alkenes with aqueous acid is generally a poor reaction in the laboratory, two indirect methods are commonly used. Hydroboration–oxidation yields the syn, non-Markovnikov hydration product, whereas oxymercuration– demercuration yields the Markovnikov hydration product (Section 5.3).