7.4 The SN1 Reaction
Most nucleophilic substitutions take place by the SN2 pathway just discussed. The reaction is favored when carried out with an unhindered substrate and a negatively charged nucleophile in a polar aprotic solvent, but is disfavored when carried out with a hindered substrate and a neutral nucleophile in a protic solvent. You might therefore expect the reaction of a tertiary substrate (hindered) with water (neutral, protic) to be among the slowest of substitution reactions. Remarkably, however, the opposite is true. The reaction of the tertiary halide 2-bromo-2-methylpropane (CH3)3CBr with H2O to give the alcohol 2-methyl-2-propanol is more than 1 million times faster than the corresponding reaction of CH3Br to give methanol.
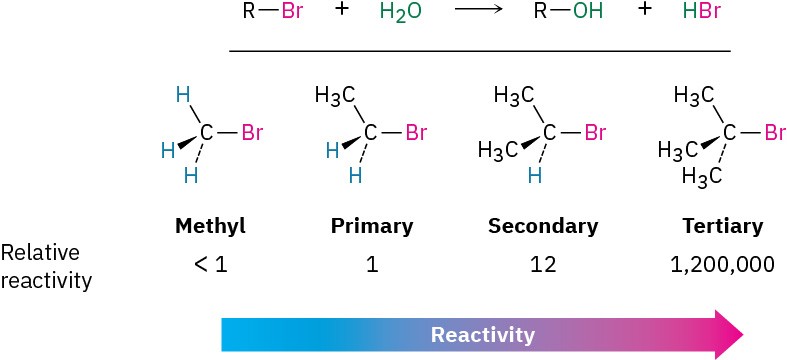 What’s going on here? A nucleophilic substitution reaction is occurring—a hydroxyl group is replacing a halogen—yet the reactivity order seems backward. These reactions can’t be taking place by the SN2 mechanism we’ve been discussing, so we must therefore conclude that they are occurring by an alternative substitution mechanism. This alternative mechanism is called the SN1 reaction, for substitution, nucleophilic, unimolecular.
What’s going on here? A nucleophilic substitution reaction is occurring—a hydroxyl group is replacing a halogen—yet the reactivity order seems backward. These reactions can’t be taking place by the SN2 mechanism we’ve been discussing, so we must therefore conclude that they are occurring by an alternative substitution mechanism. This alternative mechanism is called the SN1 reaction, for substitution, nucleophilic, unimolecular.
In contrast to the SN2 reaction of CH3Br with OH–, the SN1 reaction of (CH3)3CBr with H2O has a rate that depends only on the alkyl halide concentration and is independent of the H2O concentration. In other words, the process is a first-order reaction; the concentration of the nucleophile does not appear in the rate equation.
 To explain this result, we need to know more about kinetics measurements. Many organic reactions occur in several steps, one of which usually has a higher-energy transition state than the others and is therefore slower. We call this step with the highest transition-state energy the rate-limiting step, or rate-determining step. No reaction can proceed faster than its rate-limiting step, which acts as a kind of traffic jam, or bottleneck. In the SN1 reaction of (CH3)3CBr with H2O, the fact that the nucleophile concentration does not appear in the first-order rate equation means that it is not involved in the rate-limiting step and must therefore be involved in some other, non-rate-limiting step. The mechanism shown in Figure 7.6 accounts for these observations.
To explain this result, we need to know more about kinetics measurements. Many organic reactions occur in several steps, one of which usually has a higher-energy transition state than the others and is therefore slower. We call this step with the highest transition-state energy the rate-limiting step, or rate-determining step. No reaction can proceed faster than its rate-limiting step, which acts as a kind of traffic jam, or bottleneck. In the SN1 reaction of (CH3)3CBr with H2O, the fact that the nucleophile concentration does not appear in the first-order rate equation means that it is not involved in the rate-limiting step and must therefore be involved in some other, non-rate-limiting step. The mechanism shown in Figure 7.6 accounts for these observations.
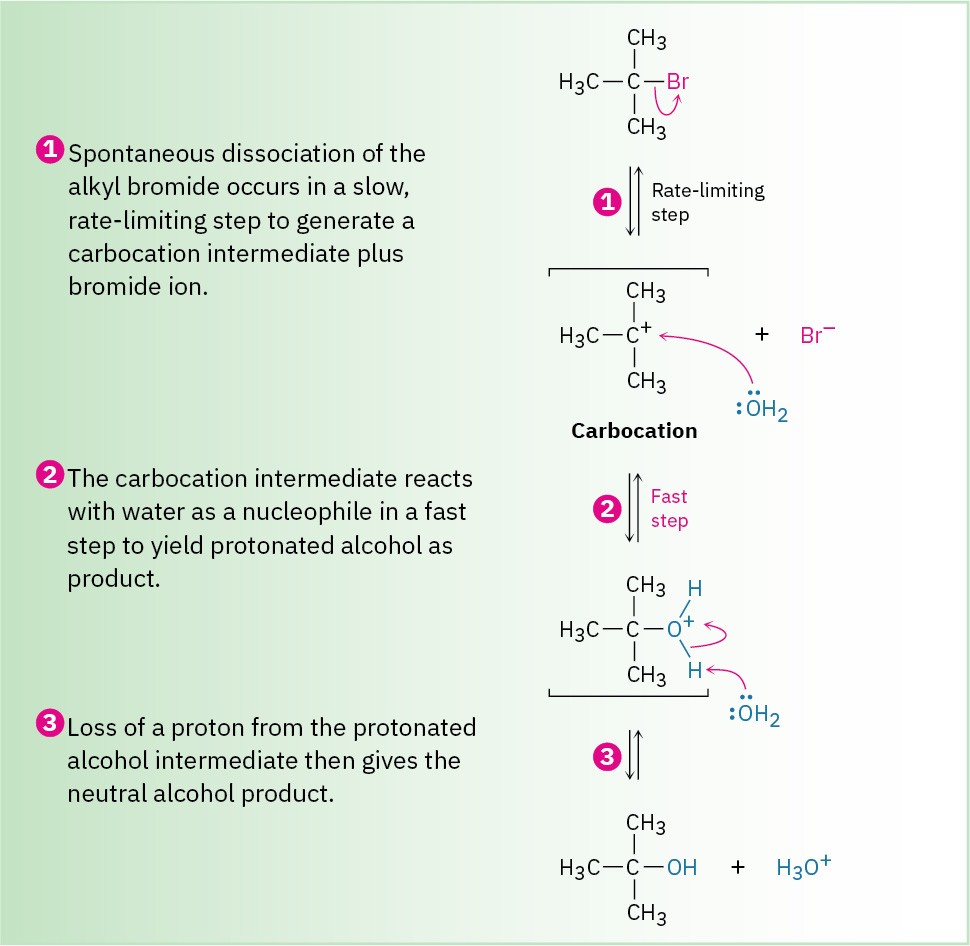
Figure 7.6 MECHANISM: The mechanism of the SN1 reaction of 2-bromo-2-methylpropane with H2O involves three steps. Step 1—the spontaneous, unimolecular dissociation of the alkyl bromide to yield a carbocation—is rate-limiting.
Unlike what occurs in an SN2 reaction, where the leaving group is displaced while the incoming nucleophile approaches, an SN1 reaction takes place by loss of the leaving group before the nucleophile approaches.
Because an SN1 reaction occurs through a carbocation intermediate, its stereochemical outcome is different from that of an SN2 reaction. Carbocations, as we’ve seen, are planar, sp2-hybridized, and achiral. Thus, if we carry out an SN1 reaction on one enantiomer of a chiral reactant and go through an achiral carbocation intermediate, the product loses its optical activity. That is, the symmetrical intermediate carbocation can react with a nucleophile equally well from either side, leading to a racemic, 50 : 50 mixture of enantiomers (Figure 7.7).
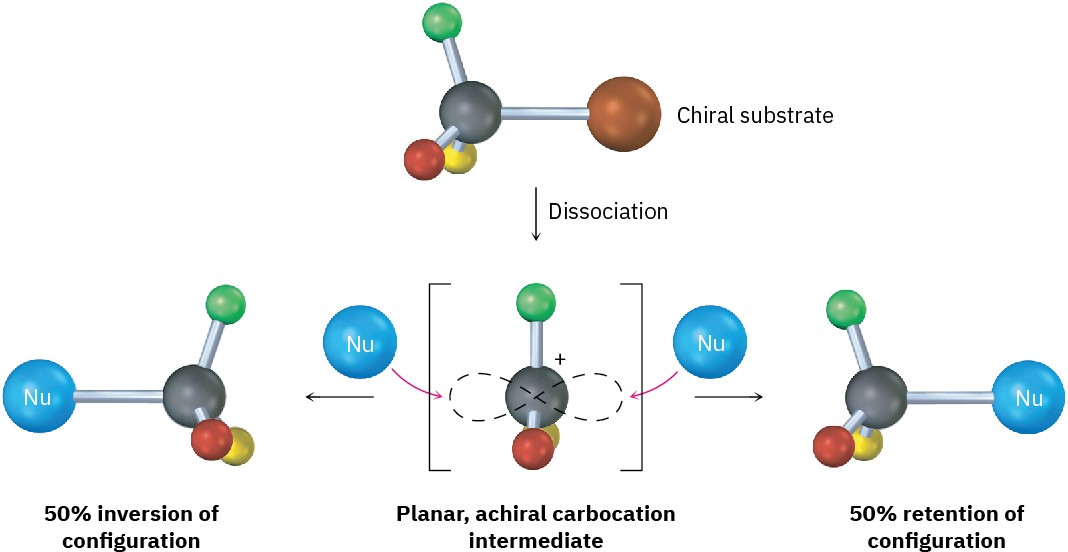 Figure 7.7 Stereochemistry of the SN1 reaction. Because the reaction goes through an achiral intermediate, an enantiomerically pure reactant gives an optically inactive racemic product.
Figure 7.7 Stereochemistry of the SN1 reaction. Because the reaction goes through an achiral intermediate, an enantiomerically pure reactant gives an optically inactive racemic product.
Problem 7.7
What product(s) would you expect from reaction of (S)-3-chloro-3-methyloctane with acetic acid? Show the stereochemistry of both reactant and product.
Problem 7.8
Assign configuration to the following substrate, and show the stereochemistry and identity of the product you would obtain by SN1 reaction with water (reddish brown = Br):