Chemistry Matters — Alkaloids: From Cocaine to Dental Anesthetics
Just as ammonia (NH3) is a weak base, there are a large number of nitrogen-containing organic compounds called amines that are also weak bases. In the early days of organic chemistry, basic amines derived from natural sources were known as vegetable alkali, but they are now called alkaloids. More than 20,000 alkaloids are known. Their study provided much of the impetus for the growth of organic chemistry in the nineteenth century and remains today an active and fascinating area of research.

Figure 2.9 The coca bush Erythroxylon coca, native to upland rain forest areas of Colombia, Ecuador, Peru, Bolivia, and western Brazil, is the source of the alkaloid cocaine. (credit: “Erythroxylum coca” by Danna Guevara/Wikimedia Commons, CC BY 4.0)
Many alkaloids have pronounced biological properties, and approximately 50% of pharmaceutical agents used today are derived from naturally occurring amines. As just three examples, morphine, an analgesic agent (painkiller), is obtained from the opium poppy Papaver somniferum. Ephedrine, a bronchodilator, decongestant, and appetite suppressant, is obtained from Ephedra sinica, an evergreen shrub native to Mongolia and northeastern China. Cocaine, both an anesthetic and a stimulant, is obtained from the coca bush Erythroxylon coca, endemic to the upland rain forest areas of central South America. (And yes, there really was a small amount of cocaine in the original Coca-Cola recipe, although it was removed in 1906.)
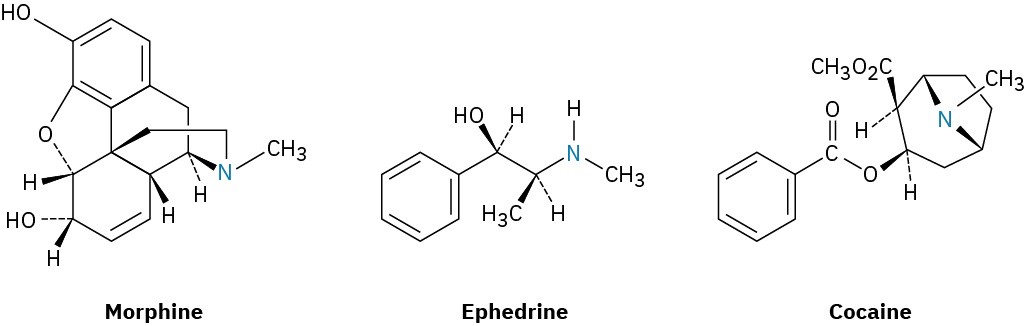
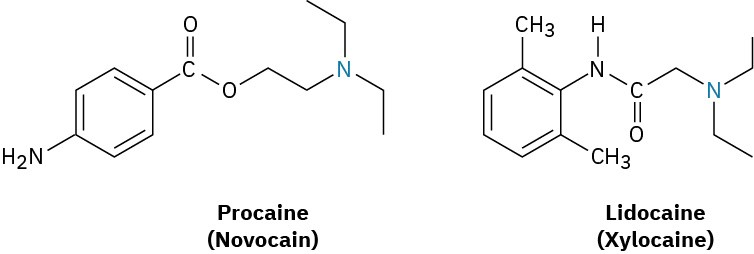
Cocaine itself is rarely used medically because it is too addictive, but its anesthetic properties provoked a long search for related but nonaddictive compounds. This search ultimately resulted in the synthesis of the “caine” anesthetics that are commonly used today in dental and surgical anesthesia. Procaine, the first such compound, was synthesized in 1898 and marketed under the name Novocain. It was rapidly adopted and remains in use today as a topical anesthetic. Other related compounds with different activity profiles followed: Lidocaine, marketed as Xylocaine, was introduced in 1943, and mepivacaine (Carbocaine) in the early 1960s. More recently, bupivacaine (Marcaine) and prilocaine (Citanest) have gained popularity. Both are quick-acting, but the effects of bupivacaine last for 3 to 6 hours while those of prilocaine fade after 45 minutes. Notice some structural similarity of all the caines to cocaine itself.
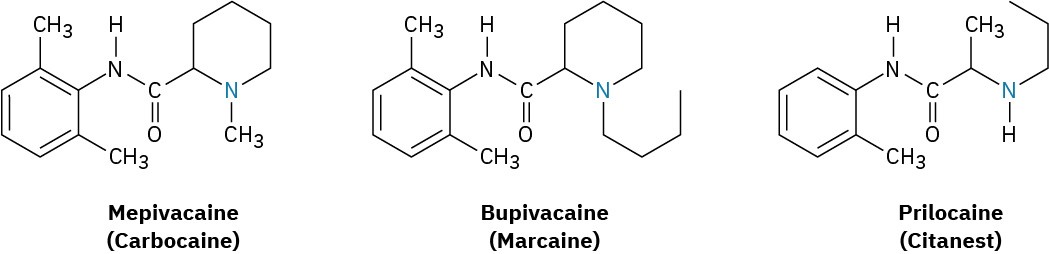
An estimate from the U.S. National Academy of Sciences is that less than 1% of all living species have been characterized. Thus, alkaloid chemistry remains an active area of research, and innumerable substances with potentially useful properties have yet to be discovered. Undoubtedly even the caine anesthetics will become obsolete at some point, perhaps supplanted by newly discovered alkaloids.

