Chemistry Matters — Vitamin C
The word vitamin, despite its common usage, is actually an imprecise term. Generally speaking, a vitamin is an organic substance that a given organism requires in small amounts to live and grow but is unable to synthesize and must obtain in its diet. Thus, to be considered a vitamin, only a small amount of the substance is needed—anywhere from a few micrograms to 100 mg or so per day. Dietary substances needed in larger amounts, such as some amino acids and unsaturated fats, are not considered vitamins.

Figure 20.8 In addition to the hazards of weather, participants in early polar expeditions often suffered from scurvy, caused by a dietary vitamin C deficiency. (credit: “Shackleton’s expedition to the Antarctic last moments of the Endurance” by Underwood & Underwood/Wikimedia Commons, Public Domain)
Furthermore, different organisms need different vitamins. More than 4000 species of mammals can synthesize ascorbic acid in their bodies, for instance, but humans are not among them. Ascorbic acid is therefore a human vitamin—what we all know as vitamin C— and must be provided by our diet. Small amounts of more than a dozen other substances are similarly required by humans: retinol (vitamin A), thiamine (vitamin B1), and tocopherol (vitamin E), for instance.
Vitamin C is surely the best known of all human vitamins. It was the first to be discovered (1912), the first to be isolated (1928), the first to be structurally characterized (1933), and the first to be synthesized in the laboratory (1933). Over 121,000 tons of vitamin C is synthesized worldwide each year, more than the total amount of all other vitamins combined. In addition to its use as a supplement, vitamin C is used as a food preservative, a “flour improver” in bakeries, and an animal-food additive.
Vitamin C is perhaps most well-known for its antiscorbutic properties, meaning that it prevents the onset of scurvy, a bleeding disease affecting those with a deficiency of fresh vegetables and citrus fruits in their diet. Sailors in the Age of Exploration were particularly susceptible to scurvy, and the death toll was high. The Portuguese explorer Vasco da Gama lost more than half his crew to scurvy during his two-year voyage around the Cape of Good Hope in 1497–1499.
More recently, large doses of vitamin C have been claimed to prevent the common cold, cure infertility, delay the onset of symptoms in acquired immunodeficiency syndrome (AIDS), and inhibit the development of gastric and cervical cancers. None of these claims have been backed by medical evidence, however. In the largest study yet of the effect of vitamin C on the common cold, a meta-analysis of more than 100 separate trials covering 40,000 people found no difference in the incidence of colds between those who took supplemental vitamin C regularly and those who did not. When taken during a cold, however, vitamin C does appear to decrease the cold’s duration by perhaps a day.
The industrial preparation of vitamin C involves an unusual blend of biological and laboratory organic chemistry, beginning with glucose and following the five-step route shown in Figure 20.9. Glucose, a pentahydroxy aldehyde, is first reduced to sorbitol, which is then oxidized by the microorganism Acetobacter suboxydans. No chemical reagent is known that is selective enough to oxidize only one of the six alcohol groups in sorbitol, so an enzymatic reaction is used. Treatment with acetone and an acid catalyst then converts four of the other hydroxyl groups into acetal linkages, and the remaining hydroxyl group is chemically oxidized to a carboxylic acid by reaction with aqueous NaOCl (household bleach). Hydrolysis with acid then removes the two acetal groups and causes an internal ester-forming reaction to give ascorbic acid. Each of the five steps has a yield better than 90%.
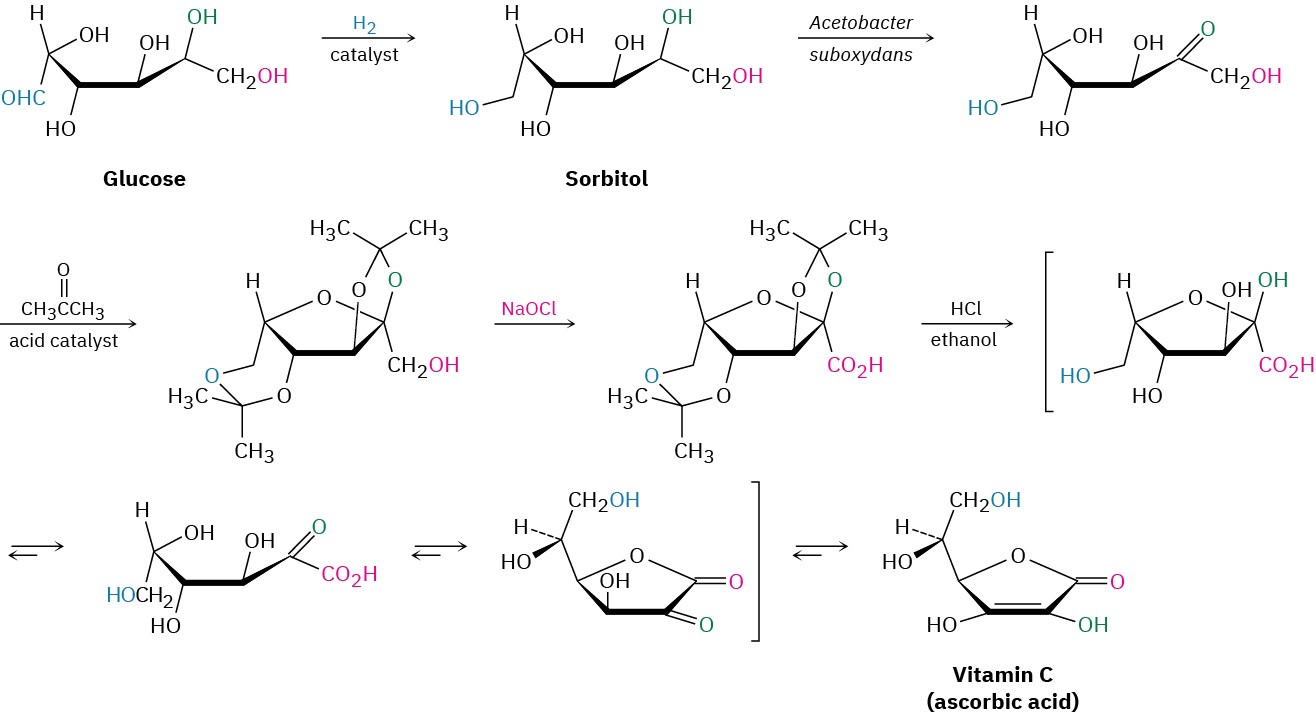
Figure 20.9The industrial synthesis of ascorbic acid from glucose.

